Degradation of mineral-immobilized pyrene by ferrate oxidation: Role of mineral type and intermediate oxidative iron species
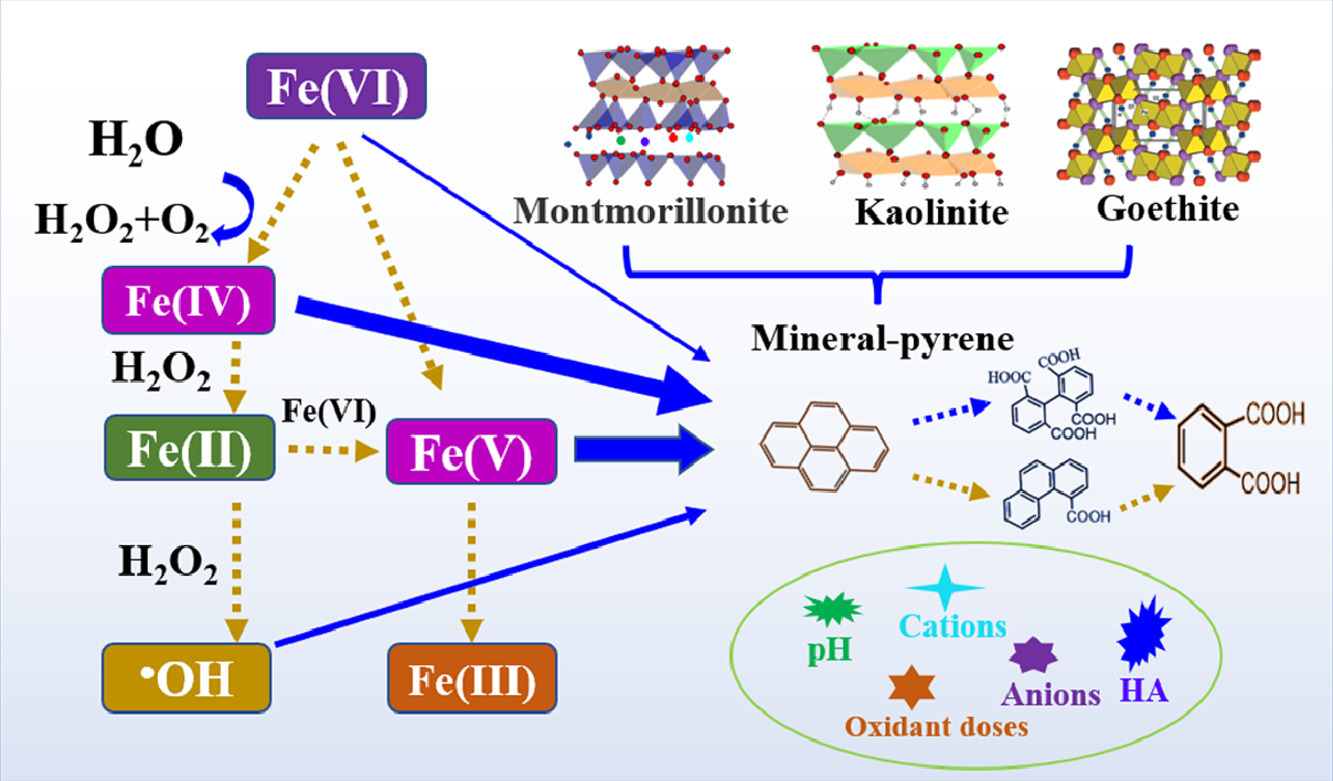
Ferrate (Fe(VI)) salts like K2FeO4 are efficient green oxidants to remediate organic contaminants in water treatment. Minerals are efficient sorbents of contaminants and also excellent solid heterogeneous catalysts which might affect Fe(VI) remediation processes. By targeting the typical polycyclic aromatic hydrocarbon compound - pyrene, the application of Fe(VI) for oxidation of pyrene immobilized on three minerals, i.e., montmorillonite, kaolinite and goethite was studied for the first time. Pyrene immobilized on the three minerals was efficiently oxidized by Fe(VI), with 87%–99% of pyrene (10 μM) being degraded at pH 9.0 in the presence of a 50-fold molar excess Fe(VI). Different minerals favored different pH optima for pyrene degradation, with pH optima from neutral to alkaline following the order of montmorillonite (pH 7.0), kaolinite (pH 8.0), and goethite (pH 9.0). Although goethite revealed the highest catalytic activity on pyrene degradation by Fe(VI), the greater noneffective loss of the oxidative species by ready self-decay in the goethite system resulted in lower degradation efficiency compared to montmorillonite. Protonation and Lewis acid on montmorillonite and goethite assisted Fe(VI) oxidation of pyrene. The intermediate ferrate species (Fe(V)/Fe(IV)) were the dominant oxidative species accountable for pyrene oxidation, while the contribution of Fe(VI) species was negligible. Hydroxyl radical was involved in mineral-immobilized pyrene degradation and contributed to 11.5%–27.4% of the pyrene degradation in montmorillonite system, followed by kaolinite (10.8%–21.4%) and goethite (5.1%–12.2%) according to the hydroxyl radical quenching experiments. Cations abundant in the matrix and dissolved humic acid hampered pyrene degradation. Finally, two different degradation pathways both producing phthalic acid were identified. The study of Professor Wang Fang and her team demonstrates efficient Fe(VI) oxidation of pyrene immobilized on minerals and contributes to the development of efficient environmentally friendly Fe(VI) based remediation techniques.

